What are electromagnetic waves and how did they discover that light is made of them? How did they discover all the electromagnetic waves (like radio waves) that we can’t even see? And how do these electromagnetic waves affect our lives? In this exceptional program, Science teacher Spiro Liacos introduces students to waves in general, and then takes a detailed and very entertaining look at radio waves, microwaves, infrared light, visible light, ultraviolet light, X-rays and gamma rays. Along the way he surfs, plays guitar and piano, flies a helicopter (a remote control one), dives into the ocean, visits the dentist, and spends some time in the tropical sun. The video comes with an accompanying worksheet and a practical activity where students measure the speed of sound. These resources are, quite simply, the easiest and the best way to teach students all about electromagnetic waves!
The preview video below contains a 6-minute excerpt of the program followed by a 2-minute trailer.
 The Question Sheet for Students:
The Question Sheet for Students:
![]() The PDF version.
The PDF version. ![]()
Google The Google Doc version. Google
Get the answers.
![]() If you have ClickView, watch the whole episode here.
If you have ClickView, watch the whole episode here.
![]() If you have Learn360, watch the whole episode here.
If you have Learn360, watch the whole episode here.
![]() If you have Films on Demand, watch the whole episode here.
If you have Films on Demand, watch the whole episode here.
![]() If you have Classroom Video, watch the whole episode here.
If you have Classroom Video, watch the whole episode here.
![]() Most of our videos are also available on SAFARI Montage. Just log in and do a quick search.
Most of our videos are also available on SAFARI Montage. Just log in and do a quick search.
![]() Don’t have any of the above? Rent or buy the Shedding Light series and/or individual programs from our Vimeo page!!
Don’t have any of the above? Rent or buy the Shedding Light series and/or individual programs from our Vimeo page!!
Speed of Sound Practical Activity (pdf, 73kB)
The Transcript (which can be used as a textbook)
Contents:
Part A: Introduction
Part B: The Discovery of Electromagnetic Waves: electricity, magnetism, their interrelation, James Maxwell, Heinrich Hertz, Guglielmo Marconi and more…
Part C: Waves: a brief look at waves where terms like frequency, wavelength, transverse and longitudinal are all explained and shown both with animations and with live footage…
Part D: Radio waves and Microwaves: the generation of radio waves and microwaves, AM and FM transmissions, microwave ovens and more…
Part E: Infrared Waves, or IR: fire, radiant heat, thermal imaging, night vision and more…
Part F: Visible Light: sunlight, lasers, colours (or colors), Hollywood special effects and the answer to the age-old question of why the sky is blue…
Part G: Ultraviolet (UV): more sunlight, ageing of the skin, UV-A, UV-B, UV-C, sunbathing, skin cancers, sunscreens and more…
Part H: X-rays: bones, X-ray photos, skeletons, dentists, tumour-blasting X-ray therapy and more…
Part I: Gamma Rays: supernovas, atoms, Geiger counters, beverage cans, bottles and more…
Part A: Introduction
 Electromagnetic Waves. Light rays are made of them. Light rays are waves of electrical and magnetic energy that travel through the air, or through the vacuum of space, or through water just to give a few examples. These light waves, these electromagnetic waves allow us to see things, because when light reflects off something and then enters our eyes, our eyes detect the light.
Electromagnetic Waves. Light rays are made of them. Light rays are waves of electrical and magnetic energy that travel through the air, or through the vacuum of space, or through water just to give a few examples. These light waves, these electromagnetic waves allow us to see things, because when light reflects off something and then enters our eyes, our eyes detect the light.
But it turns out that the light waves that we can see are not the only electromagnetic waves that exist. In fact, many electromagnetic waves are invisible to us, like the ones this wireless microphone is producing.
Radio waves, microwaves, infrared waves, visible light, which we can see, Ultraviolet rays, X-rays and Gamma rays are all electromagnetic waves. All these waves travel out, or radiate out from whatever produces them and so they’re often referred to as electromagnetic radiation or EMR. The complete range of electromagnetic waves is called the electromagnetic spectrum.
Even though they are all electromagnetic waves, Infrared and Ultraviolet waves are often referred to as infrared “light” and ultraviolet “light” (along with visible light of course), while radio waves and microwaves are usually referred to as “waves” and X-rays and gamma rays are usually called “rays”.
So what exactly are electromagnetic waves, and how did they discover that light is made of them. And how did they learn that there are many different types that we can’t even see. Well, let’s look into how they were discovered first and then examine all the different parts of the spectrum.
Part B: The Discovery of Electromagnetic Waves

 The question of what light is had been around since ancient times, but only in the 1860s did a scientist called James Clerk Maxwell come up with a really good explanation. It had already been discovered that a moving magnet in a coil of wire produces electricity. It had also been discovered that a coil of wire with electricity running through it would become magnetised.
The question of what light is had been around since ancient times, but only in the 1860s did a scientist called James Clerk Maxwell come up with a really good explanation. It had already been discovered that a moving magnet in a coil of wire produces electricity. It had also been discovered that a coil of wire with electricity running through it would become magnetised.
 This so-called electromagnet isn’t all that strong, but this one here, in this metal recycling yard, which is being powered by a very powerful generator, can lift up huge amounts of steel. When the electricity is turned off, the magnetism stops and the metal falls away. Electricity and magnetism are very much related. Surrounding a magnet is an invisible, so-called magnetic field, which can apply a force on things.
This so-called electromagnet isn’t all that strong, but this one here, in this metal recycling yard, which is being powered by a very powerful generator, can lift up huge amounts of steel. When the electricity is turned off, the magnetism stops and the metal falls away. Electricity and magnetism are very much related. Surrounding a magnet is an invisible, so-called magnetic field, which can apply a force on things.
 An electric field is similar. Here for example, I can rub a balloon to charge it with static electricity. The static electricity produces an invisible electric field which applies a force on these pieces of styrofoam. The balloon acquires a negative charge and the hair gains a positive charge.
An electric field is similar. Here for example, I can rub a balloon to charge it with static electricity. The static electricity produces an invisible electric field which applies a force on these pieces of styrofoam. The balloon acquires a negative charge and the hair gains a positive charge.

 Now scientists in the 1800s carried out lots of experiments to do with electricity and magnetism where they actually measured things like for example how the amount of electricity flowing in an electromagnet affects its strength.
Now scientists in the 1800s carried out lots of experiments to do with electricity and magnetism where they actually measured things like for example how the amount of electricity flowing in an electromagnet affects its strength.
James Maxwell used the results to come up with these four mathematical equations which linked all the data together. Now stay with me for a moment. The symbols all mean something; the B for example refers to how strong the magnetic field is. Maxwell was able to kind of read the equations, like a sentence almost, and they told him that whenever a charged particle accelerates, an electric-field wave and a magnetic-field wave are created.
He worked out that these two waves are produced at right angles to each other and that they’re inseparable, so they effectively form a single electromagnetic wave which, once created, travels on forever, or at least until it hits something.
Maxwell used his equations to calculate that the speed these electromagnetic waves travel at was equal to the speed of light, so he proposed that light must be made of electromagnetic waves. (The speed of light had been measured a few years earlier by French Scientists Hippolyte Fizeau and Léon Foucault.)
Maxwell also suggested that it wasn’t just visible light that is made of these waves. There were likely to be electromagnetic waves which we couldn’t see, and which were waiting to be discovered.

 About twenty years later, in 1888, a scientist called Heinrich Hertz used Maxwell’s equations to design a transmitter and a receiver of what we now call radio waves. It worked in a very similar way to this induction coil and spark generator. It moves electrons back and forth really fast along here and creates sparks in this small gap . The accelerating electrons produce electromagnetic waves.
About twenty years later, in 1888, a scientist called Heinrich Hertz used Maxwell’s equations to design a transmitter and a receiver of what we now call radio waves. It worked in a very similar way to this induction coil and spark generator. It moves electrons back and forth really fast along here and creates sparks in this small gap . The accelerating electrons produce electromagnetic waves.
Atoms are made of a central nucleus which is surrounded by electrons. The nucleus is made up of positively charged protons and neutral neutrons, so it has a positive charge. The electrons have a negative charge. In metals, the outermost electrons are free to move from atom to atom. Electricity is, basically, the movement of these electrons along a conductor, when a voltage is applied across it. At really high voltages, electrons can jump across a gap in a wire, creating a spark.
 Near his spark generator, Hertz set up a loop of wire with a tiny gap in it. Some of the waves being produced by the spark generator were absorbed by the loop of wire and this caused tiny sparks to be produced between the gap in the loop. The waves were invisible, but the loop of wire detected them.
Near his spark generator, Hertz set up a loop of wire with a tiny gap in it. Some of the waves being produced by the spark generator were absorbed by the loop of wire and this caused tiny sparks to be produced between the gap in the loop. The waves were invisible, but the loop of wire detected them.
This experiment was the first real proof that invisible electromagnetic waves existed. Up until then, they were just a theory based on Maxwell’s equations. It goes to show just how powerful mathematics can be.
I can detect the radio waves produced by this spark generator using this AM radio.
I’ll use a switch to turn the sparks on and off, and I’ll bring a microphone up close so that you hear the different sounds being produced. The radio’s antenna picks up the radio waves and you can hear the so-called static through the speaker.
When people asked Hertz what these invisible electromagnetic waves were useful for, Hertz answered that they were of no use whatsoever. The experiment just proved that Maxwell was right, he said, and that these invisible-to-the-eye electromagnetic waves exist.
But within a few years, in the 1890s, Italian scientist Guglielmo Marconi invented a way of reliably sending and receiving these waves over long distances, bringing in the new era of radio communications.
The first radio transmitters were, basically, just powerful spark generators and the receivers just made a buzzing sound so initially, messages could only be sent by Morse Code, but things obviously got a lot better.
You can experiment at home with a piezoelectric oven igniter. These devices also produce radio waves every time they produce a spark.
Now before we delve into electromagnetic waves more deeply, it’ll probably help our understanding of these waves if we take a brief look at waves in general, so let’s do that right now.
Part C: Waves
Waves are disturbances that carry energy from one place to another. There are many different types of waves. For example,

 Slinky waves… sound waves… water waves… and seismic waves, the waves generated in the ground by earthquakes. All of these waves are called mechanical waves, because they need what’s called a medium to travel through. In a slinky wave, for example, the slinky is the medium. The movement of one coil makes the next one move, which makes the next one move and so on. This basic process occurs in all mechanical waves.
Slinky waves… sound waves… water waves… and seismic waves, the waves generated in the ground by earthquakes. All of these waves are called mechanical waves, because they need what’s called a medium to travel through. In a slinky wave, for example, the slinky is the medium. The movement of one coil makes the next one move, which makes the next one move and so on. This basic process occurs in all mechanical waves.
 When each coil moves perpendicular to the direction of the wave, the wave is called a transverse wave. Each loop is moving only up and down as the wave moves from left to right. You can see that the blue tape on one of the coils does more or less the same thing. The waves move sideways, but each particle moves only up and down. One of Microsoft Powerpoint’s animations is called “wave”, and it creates a transverse wave by moving each letter up and down.
When each coil moves perpendicular to the direction of the wave, the wave is called a transverse wave. Each loop is moving only up and down as the wave moves from left to right. You can see that the blue tape on one of the coils does more or less the same thing. The waves move sideways, but each particle moves only up and down. One of Microsoft Powerpoint’s animations is called “wave”, and it creates a transverse wave by moving each letter up and down.
 When each coil moves side to side in the same direction that the wave is travelling in, the wave is called a longitudinal wave. Each loop is moving first to the right and then to the left as the wave moves from left to right. Sound waves are longitudinal waves. Electromagnetic Waves are different from all other types of waves in that they don’t need a medium to travel through. The light coming from the sun for example, can get to us here on earth even though the space between the earth and the sun is a vacuum. Light doesn’t need a medium because it’s a self-contained wave of electrical and magnetic energy.
When each coil moves side to side in the same direction that the wave is travelling in, the wave is called a longitudinal wave. Each loop is moving first to the right and then to the left as the wave moves from left to right. Sound waves are longitudinal waves. Electromagnetic Waves are different from all other types of waves in that they don’t need a medium to travel through. The light coming from the sun for example, can get to us here on earth even though the space between the earth and the sun is a vacuum. Light doesn’t need a medium because it’s a self-contained wave of electrical and magnetic energy.
 Sound waves, seismic waves and slinky waves are all kind of similar. Electromagnetic waves are in a class of their own. We can represent a wave with a simple diagram. We’ll use a transverse wave because it’s so much easier to picture. Most waves in nature have a positive and a negative component. The top of the wave is called a crest, while the bottom of the wave is called a trough.The so-called wavelength of a wave is the distance from one crest to the next, or from one trough to the next. This distance is also the wavelength. Wavelength is usually measured in metres. The Greek letter lambda, which is the Greek letter l, is used as the symbol for wavelength. So, if the wavelength was 2 metres, I would label this diagram by writing ? = 2m, but I would read this as: wavelength = 2 metres.A wave’s amplitude is the height of the crest of the wave, or, if you like, the depth of the trough.When two waves meet, they pass straight through each other.
Sound waves, seismic waves and slinky waves are all kind of similar. Electromagnetic waves are in a class of their own. We can represent a wave with a simple diagram. We’ll use a transverse wave because it’s so much easier to picture. Most waves in nature have a positive and a negative component. The top of the wave is called a crest, while the bottom of the wave is called a trough.The so-called wavelength of a wave is the distance from one crest to the next, or from one trough to the next. This distance is also the wavelength. Wavelength is usually measured in metres. The Greek letter lambda, which is the Greek letter l, is used as the symbol for wavelength. So, if the wavelength was 2 metres, I would label this diagram by writing ? = 2m, but I would read this as: wavelength = 2 metres.A wave’s amplitude is the height of the crest of the wave, or, if you like, the depth of the trough.When two waves meet, they pass straight through each other.
Here a larger-amplitude wave pulse moving towards the right meets a smaller-amplitude wave pulse moving towards the left.



After they pass through each other, the larger wave pulse is still moving towards the right and the smaller wave pulse continues moving towards the left.
If waves didn’t pass through each other, then, for example, all the light reflecting off me towards the camera would crash into all the light reflecting off the camera towards me. The waves would bounce off each other and scatter in every direction. All we would see is a blur of light.

 At the point where the waves meet, the amplitudes of the waves add up. This is called superposition. This amplitude plus this amplitude equals this amplitude when the two waves superimpose.
At the point where the waves meet, the amplitudes of the waves add up. This is called superposition. This amplitude plus this amplitude equals this amplitude when the two waves superimpose.
Superposition is what causes the colour in bubbles. The light reflecting of the top surface superimposes onto the light reflecting off the bottom surface. The two waves get stronger and that particular wavelength becomes the colour of the film.
 If the wave pulses are on the opposite side of the slinky, the waves superimpose to temporarily reduce in overall amplitude, before moving on. DVD players use this principle. They shine a laser beam onto the disc, and the way the laser reflects off the tiny pits embedded in the disc causes the light to either appear bright at the sensor or to appear dim.
If the wave pulses are on the opposite side of the slinky, the waves superimpose to temporarily reduce in overall amplitude, before moving on. DVD players use this principle. They shine a laser beam onto the disc, and the way the laser reflects off the tiny pits embedded in the disc causes the light to either appear bright at the sensor or to appear dim.
Frequency, given the symbol f, is the number of complete wavelengths that pass a given point per second. It’s measured in Hertz (Hz).
The frequency of the top wave is 1Hz; 1 complete wavelength is being produced per second, while the frequency of the bottom wave is 2Hz.
Here, I’ve attached this speaker to a signal generator. I’ve set the output to 1Hz, so the speaker is vibrating once per second. I can increase the frequency to 2Hz, that is 2 vibrations per second, and then to 3 Hz and so on. Humans can hear sounds only if they’re above about 20 Hz. This is 60Hz, 90Hz, 400Hz, 800Hz, about 9000Hz. Humans can hear only up to about 20,000Hz. My ears are starting to Hertz.
 That sounds better. Middle C has a frequency of 262 Hz, The A note above it 440Hz, and this C 524Hz. The lowest C on an 88-key piano has a frequency of 33Hz, while the highest C has a frequency of 4186Hz.
That sounds better. Middle C has a frequency of 262 Hz, The A note above it 440Hz, and this C 524Hz. The lowest C on an 88-key piano has a frequency of 33Hz, while the highest C has a frequency of 4186Hz.
 Wave speed is a measure of how fast a wave moves. The speed of something is often expressed in km/hr or in miles per hour, but scientists usually work in metres per second. We can use an equation to calculate speed. Average speed = distance / time. I can run 100metres in about 15 seconds, so my average speed over this distance is 100m over 15 seconds which equals 6.7m/s.
Wave speed is a measure of how fast a wave moves. The speed of something is often expressed in km/hr or in miles per hour, but scientists usually work in metres per second. We can use an equation to calculate speed. Average speed = distance / time. I can run 100metres in about 15 seconds, so my average speed over this distance is 100m over 15 seconds which equals 6.7m/s.
Light travels at 300,000,000 metres per second, while sound travels at only about 340m/s. You can tell the difference when watching a starter’s pistol go off from a distance, in this case 100metres. You see the smoke come out first and then hear the sound. Let’s watch it again….
It’s even more noticeable when the distance increases. This field is 300 metre long. If I stand at one end and film from the other, the sound takes nearly a whole second to reach the camera’s microphone. If speed = distance over time, it follows that distance = speed x time and that time = distance over speed. The time it takes the light from the smoke to travel 300m at a speed of 300,000,000m/s is only 0.000,0001 seconds. Practically no time at all.

 The sound wave though, travelling at only about 340 m/s takes about 0.88 seconds to reach the camera’s microphone. Let’s watch one more time. You’ll see the smoke first and then hear the sound about 1 second later. It turns out that in any given medium, a wave will travel at a constant speed, regardless of its frequency. Low frequency sound waves travel at the same speed in air as high frequency sound waves: about 340m/s. And, soft sounds with small amplitudes, travel at the same speed as loud sounds with large amplitudes. If they didn’t all travel at the same speed, some sounds at say a concert would reach you before other sounds. The speed of sound only changes if the temperature or the pressure of the air change. In 0 degrees Celsius air, sound travels at 331m/s no matter what its frequency is, while in 20 degrees Celsius air, it travels at 343m/s. It travels at different speeds in different substances. In a vacuum, all electromagnetic waves travel at 300, 000km/s (300,000,000m/s) whether they’re low frequency radio waves with frequencies of around 10,000Hz, or high frequency gamma rays with frequencies of around 10 to the 20 Hertz. However, though changing the frequency doesn’t affect the speed of a wave, it does affect the wavelength. The two waves that we looked at before are travelling at the same speed, but the wavelength of the 1Hz wave is twice as long as the wavelength of the 2Hz wave.
The sound wave though, travelling at only about 340 m/s takes about 0.88 seconds to reach the camera’s microphone. Let’s watch one more time. You’ll see the smoke first and then hear the sound about 1 second later. It turns out that in any given medium, a wave will travel at a constant speed, regardless of its frequency. Low frequency sound waves travel at the same speed in air as high frequency sound waves: about 340m/s. And, soft sounds with small amplitudes, travel at the same speed as loud sounds with large amplitudes. If they didn’t all travel at the same speed, some sounds at say a concert would reach you before other sounds. The speed of sound only changes if the temperature or the pressure of the air change. In 0 degrees Celsius air, sound travels at 331m/s no matter what its frequency is, while in 20 degrees Celsius air, it travels at 343m/s. It travels at different speeds in different substances. In a vacuum, all electromagnetic waves travel at 300, 000km/s (300,000,000m/s) whether they’re low frequency radio waves with frequencies of around 10,000Hz, or high frequency gamma rays with frequencies of around 10 to the 20 Hertz. However, though changing the frequency doesn’t affect the speed of a wave, it does affect the wavelength. The two waves that we looked at before are travelling at the same speed, but the wavelength of the 1Hz wave is twice as long as the wavelength of the 2Hz wave.
 It’s a bit like firing a gun. It doesn’t matter how frequently you pull the trigger, the bullets always fly out at the same speed. If you fire one shot per second, the bullets will be a certain distance apart. If you fire two shots per second, the bullets will travel at the same speed, but they’ll be closer together. Similarly, a higher frequency wave has a shorter wavelength. If you fire a gun with a really high frequency, the bullets will be really close together.
It’s a bit like firing a gun. It doesn’t matter how frequently you pull the trigger, the bullets always fly out at the same speed. If you fire one shot per second, the bullets will be a certain distance apart. If you fire two shots per second, the bullets will travel at the same speed, but they’ll be closer together. Similarly, a higher frequency wave has a shorter wavelength. If you fire a gun with a really high frequency, the bullets will be really close together.
There’s a really simple equation, called the wave equation that ties wave speed, frequency and wavelength all together: wave speed = frequency x wavelength.
If for example you were producing waves with a frequency of 3 Hz, that is 3 individual waves per second, and the wavelength for each one was 2 metres, then it follows that the front of the original wave is 6 metres away after 1 second, so it must be travelling at 6m/s. wave speed = frequency x wavelength. It can’t be anything else.
 So, if you know any two of these three values, you can calculate the third value by using the wave equation. Since the speed of a wave in any given medium stays the same, if the frequency increases, the wavelength has to decrease, and vice versa. Low frequency radio waves, have long wavelengths of more than a metre, while high frequency gamma rays have really short wavelengths, of around 1 trillionth of a metre.
So, if you know any two of these three values, you can calculate the third value by using the wave equation. Since the speed of a wave in any given medium stays the same, if the frequency increases, the wavelength has to decrease, and vice versa. Low frequency radio waves, have long wavelengths of more than a metre, while high frequency gamma rays have really short wavelengths, of around 1 trillionth of a metre.
To express really large and really small quantities, scientists often use standard form or various prefixes.
One picometre for example = 10-12m that is 1 trillionth of a metre or one millionth millionth. It’s very difficult to picture anything this small, but for what it’s worth, the smallest atoms, which are Helium atoms have a diameter of about 60 picometres. (16.67 million can fit across one millimetre)
1 trillion millimetres = 1 billion metres = 1 million kilometres. The moon is about 378,000km away, so 1 trillion millimetres is a distance 3 times the Earth-Moon distance. I still can’t fully picture it!
 We then have one nanometre = 10-9m; 1 billionth of a metre (one thousandth millionth) and One micrometre (or micrometre) = 10-6m; 1 millionth of a metre. The little symbol here (?), pronounced mu, is the Greek letter m. One micrometre is often called one micron. Human hair has a diameter of about 60 to 80 micrometres. Millimetres and Kilometres are fairly well known. One millimetre = 10-3m; 1 thousandth of a metre. One kilometre = 103m; 1 thousand metres. Now I could say one megametre next, but it’s not an expression that really gets used.
We then have one nanometre = 10-9m; 1 billionth of a metre (one thousandth millionth) and One micrometre (or micrometre) = 10-6m; 1 millionth of a metre. The little symbol here (?), pronounced mu, is the Greek letter m. One micrometre is often called one micron. Human hair has a diameter of about 60 to 80 micrometres. Millimetres and Kilometres are fairly well known. One millimetre = 10-3m; 1 thousandth of a metre. One kilometre = 103m; 1 thousand metres. Now I could say one megametre next, but it’s not an expression that really gets used.
However, Megahertz is an expression that gets used. One Megahertz = 106Hz or 1 million Hertz.
We then get One Gigahertz = 109Hz; one billion Hertz.
One Terahertz = 1012Hz; one trillion Hertz.
and one Petahertz = 1015 Hz; one thousand trillion Hertz.
Since electromagnetic waves differ enormously in frequency and wavelength, scientists have classified them into broad groups. So let’s now take a look at these groups, starting with radio waves and microwaves.
Part D: Radio waves and Microwaves.
Radio waves have the lowest frequency of all electromagnetic waves. They typically range from a few tens of thousands of Hertz right up to 300 Megahertz, that is, 300 million Hertz, which corresponds to wavelengths of 10s of kilometres down to 1metre.
Microwaves have frequencies of between 300Megahertz and 300Gighertz, with wavelengths ranging from 1m down to 1mm.
Radio waves and microwaves are used in communications. Even though most people associate microwaves with microwave ovens, microwaves of various frequencies actually play a huge role in global and local information networks.
 Radio waves and microwaves are generated by electrical circuits which make the electrons in a transmitting antenna (or transmitter) move up and down really fast. This one is producing microwaves with a frequency of 2.4 GHz (that is 2.4 billion hertz). The receiving antenna and the electronic circuits in the helicopter are tuned to this frequency.
Radio waves and microwaves are generated by electrical circuits which make the electrons in a transmitting antenna (or transmitter) move up and down really fast. This one is producing microwaves with a frequency of 2.4 GHz (that is 2.4 billion hertz). The receiving antenna and the electronic circuits in the helicopter are tuned to this frequency.
Radio stations use powerful transmitters, usually placed on tall buildings or on hill tops, to broadcast radio waves long distances.
AM radio stations transmit in frequencies ranging from about 500kiloHertz to 1700kiloHertz. TEXT: 1kiloHertz is a thousand Hertz.
FM stations transmit in frequencies ranging from about 88MegaHertz to about 108MegaHertz. (TEXT) 1 MegaHertz equals 1 million Hertz.
The frequencies are marked on radios.
To transmit the sound of someone talking at a radio station, a microphone is used to convert the sound wave into an electrical signal, which is usually called the audio signal.
 I can show you what an audio signal looks like with this microphone and this Cathode-ray oscilloscope. The audio signal exactly matches the pattern of vibrations produced by the thing making the sound, in this case my vocal cords and mouth. I can also use audio-editing software to show you what an audio signal looks like
I can show you what an audio signal looks like with this microphone and this Cathode-ray oscilloscope. The audio signal exactly matches the pattern of vibrations produced by the thing making the sound, in this case my vocal cords and mouth. I can also use audio-editing software to show you what an audio signal looks like
So how is the audio signal transmitted? It’s transmitted using what’s called a carrier wave. Every radio station uses its own carrier wave set to a specific frequency.
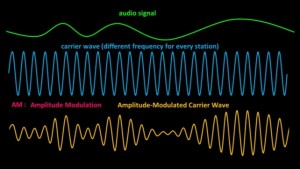 The audio signal, that might look something like this, is combined with the carrier wave which we’ll show like this. though in reality its frequency would be much much higher when compared with the audio signal. In AM transmissions, the carrier wave undergoes what’s called amplitude modulation. To modulate something means to change it in some way.
The audio signal, that might look something like this, is combined with the carrier wave which we’ll show like this. though in reality its frequency would be much much higher when compared with the audio signal. In AM transmissions, the carrier wave undergoes what’s called amplitude modulation. To modulate something means to change it in some way.
 The electrical circuits of the transmitter modulate the amplitude of the carrier wave to copy the audio signal. The frequency and wavelength of the modulated wave don’t change. Only its amplitude does so that it exactly matches the audio signal. What comes out of the antenna is a radio wave kind of like this.
The electrical circuits of the transmitter modulate the amplitude of the carrier wave to copy the audio signal. The frequency and wavelength of the modulated wave don’t change. Only its amplitude does so that it exactly matches the audio signal. What comes out of the antenna is a radio wave kind of like this.
The amplitude-modulated carrier wave is absorbed by the antenna of a radio, making the electrons in it vibrate up and down if the radio is tuned to that frequency. The electronic circuits in the radio filter out the carrier wave, but they keep its shape. This signal is amplified and sent to the speakers which then vibrate with the same pattern that the announcer’s voice originally did.
 FM stands for frequency modulation. If the audio signal looks like this, the frequency of an FM radio stations’s carrier wave is modulated to match it. The frequency of the carrier wave is increased for the peaks of the audio signal and is decreased for the troughs. However, the modulation shown here is hugely exaggerated. Absolute Radio, in London, UK, uses a 105.8MegaHertz carrier wave, which is continuously modulated by only about 0.1% (plus or minus 75kHz). But that’s enough for the electronics in the radio to recreate the audio signal which is then amplified and sent to the speakers.
FM stands for frequency modulation. If the audio signal looks like this, the frequency of an FM radio stations’s carrier wave is modulated to match it. The frequency of the carrier wave is increased for the peaks of the audio signal and is decreased for the troughs. However, the modulation shown here is hugely exaggerated. Absolute Radio, in London, UK, uses a 105.8MegaHertz carrier wave, which is continuously modulated by only about 0.1% (plus or minus 75kHz). But that’s enough for the electronics in the radio to recreate the audio signal which is then amplified and sent to the speakers.
(The amplitude and frequency of an audio signal typically change very rapidly, but since humans can hear frequencies of only between 20Hz and 20,000Hz, that’s also the frequency range of a typical audio signal. However, as we’ve seen, FM radio stations use carrier waves which have frequencies of about 100MHz, so the amount of modulation needed is tiny.)
The frequency of a radio station’s carrier wave is often mentioned by the presenters and can often be seen on the station’s logo.
AM and FM radio are only two of the bands within the electromagnetic spectrum that are used for communications. There are many more. For example, TV stations and walkie talkies transmit radio waves, while mobile phones, Bluetooth devices and Ku-band satellites use microwaves. There are literally hundreds of other examples.
 This transmitter is transmitting microwaves with a frequency of 626.3MHz. Some transmissions, like AM and FM radio broadcasts, are analog, where the signal continuously changes over a range of values, while other transmissions are digital, where short pulses are sent in quick succession, millions of them per second.
This transmitter is transmitting microwaves with a frequency of 626.3MHz. Some transmissions, like AM and FM radio broadcasts, are analog, where the signal continuously changes over a range of values, while other transmissions are digital, where short pulses are sent in quick succession, millions of them per second.
Television stations transmit their signals at frequencies ranging from about 5MHz to about 9MHz. They used to use FM for the sound and AM for the picture, but by the early 2010s most countries had adopted digital transmissions. Instead of a modulated wave, TV stations now broadcast their radio waves in pulses, millions per second, at the frequency that they’ve been allocated.
Microwave ovens generate microwaves that have a frequency of about 2.45GigaHertz and a wavelength of about 12cm.
 When microwaves of this particular frequency enter food, they cause the water molecules in the food to vibrate faster and they get hotter. Water molecules, made up of one oxygen atom bonded to two hydrogen atoms are slightly negatively charged on one side and slightly positively charged on the other side. As the microwaves pass through the food, the slightly charged ends of the water molecules are pushed back and forth by the waves’ electric and magnetic fields. The energy of the microwaves is therefore absorbed by the water molecules, they vibrate faster, and their temperature increases. They then bump into other molecules in the food and heat them up as well.
When microwaves of this particular frequency enter food, they cause the water molecules in the food to vibrate faster and they get hotter. Water molecules, made up of one oxygen atom bonded to two hydrogen atoms are slightly negatively charged on one side and slightly positively charged on the other side. As the microwaves pass through the food, the slightly charged ends of the water molecules are pushed back and forth by the waves’ electric and magnetic fields. The energy of the microwaves is therefore absorbed by the water molecules, they vibrate faster, and their temperature increases. They then bump into other molecules in the food and heat them up as well.

 Fat and sugar molecules are also heated directly by microwaves but to a much lesser extent. Most plastic and ceramic containers don’t absorb these microwaves, so they only get hot if they’re in contact with the food that is being been heated.
Fat and sugar molecules are also heated directly by microwaves but to a much lesser extent. Most plastic and ceramic containers don’t absorb these microwaves, so they only get hot if they’re in contact with the food that is being been heated.
If we fill one plastic container with water and leave another one empty, and place them both into the microwave oven, we can see the difference.
After two minutes, this container has stayed cool, because it hasn’t absorbed any microwaves. The water that has been heated up in this container though, has made this container very hot.
Above frequencies of 300GHz, we enter the so-called infrared region. So let’s now take a look at, or at least feel the warmth of, infrared light.
Part E: Infrared Radiation (or Infrared Light) or Infrared Waves, or IR
Infrared waves have frequencies that range from 300Gigahertz to 430terahertz, that is, 430trillion Hertz. These frequencies correspond to wavelengths of between 1mm and 700 nanometres. Infrared light, is generated by the vibrations of atoms and their electrons. As atoms vibrate, which they’re doing constantly, they release electromagnetic waves, mostly infrared.

 This fire is generating a little visible light, but it’s generating far more infrared light, which I can’t see with my eyes, but which I can feel as heat on my skin. Infrared light is therefore often called thermal radiation or radiant heat. Once the fire has burned down a little, we can use the radiant heat coming from the hot coals to cook meat. The hot coals are also warming the air, but it’s not the hot air that’s cooking the meat, it’s the infrared radiation. A toaster also uses infrared light to toast bread.
This fire is generating a little visible light, but it’s generating far more infrared light, which I can’t see with my eyes, but which I can feel as heat on my skin. Infrared light is therefore often called thermal radiation or radiant heat. Once the fire has burned down a little, we can use the radiant heat coming from the hot coals to cook meat. The hot coals are also warming the air, but it’s not the hot air that’s cooking the meat, it’s the infrared radiation. A toaster also uses infrared light to toast bread.
 While this fire has died down it’s still giving off an incredible amount of infrared light energy. So just imagine how much radiant heat is produced in a bush fire. Fire fighters and news reporters, in this case from Channel 7 in Sydney, have to be very careful because radiant heat alone coming from a wall of burning trees can cause severe burns even if the actual flames of the fire are some distance away. TV stations often use helicopters to bring us vision of large fires, and Fire fighters always wear protective clothing to protect themselves from radiant heat. This sausage didn’t come anywhere near an actual flame. Radiant heat did this. Of course apart from the flames and the radiant heat, the smoke can also be quite deadly.
While this fire has died down it’s still giving off an incredible amount of infrared light energy. So just imagine how much radiant heat is produced in a bush fire. Fire fighters and news reporters, in this case from Channel 7 in Sydney, have to be very careful because radiant heat alone coming from a wall of burning trees can cause severe burns even if the actual flames of the fire are some distance away. TV stations often use helicopters to bring us vision of large fires, and Fire fighters always wear protective clothing to protect themselves from radiant heat. This sausage didn’t come anywhere near an actual flame. Radiant heat did this. Of course apart from the flames and the radiant heat, the smoke can also be quite deadly.
 Now infrared light is not just given off by things that we would normally call hot. It turns out that everything gives off infrared light, including you and me, because everything is made of vibrating atoms. Special Thermal imaging cameras can be used to detect the infrared light being emitted from things.
Now infrared light is not just given off by things that we would normally call hot. It turns out that everything gives off infrared light, including you and me, because everything is made of vibrating atoms. Special Thermal imaging cameras can be used to detect the infrared light being emitted from things.
 Colder objects produce a range of lower frequency waves because the atoms and their electrons are vibrating slower, while warmer objects produce higher frequency waves, and more of them, because the atoms and their electrons are vibrating faster. So, in other words, if you can determine what infrared frequencies an object is emitting you can tell its temperature.
Colder objects produce a range of lower frequency waves because the atoms and their electrons are vibrating slower, while warmer objects produce higher frequency waves, and more of them, because the atoms and their electrons are vibrating faster. So, in other words, if you can determine what infrared frequencies an object is emitting you can tell its temperature.
 A thermal imaging camera does exactly that. The electronic sensors inside the camera can distinguish between the different frequencies of infrared light being emitted, and so it can tell what temperature something is. It then displays what’s called a false-colour image, which shows the object’s temperature. Different temperatures are represented on the screen by different colours. Some people prefer the rainbow palette, while others prefer the ironbow palette.
A thermal imaging camera does exactly that. The electronic sensors inside the camera can distinguish between the different frequencies of infrared light being emitted, and so it can tell what temperature something is. It then displays what’s called a false-colour image, which shows the object’s temperature. Different temperatures are represented on the screen by different colours. Some people prefer the rainbow palette, while others prefer the ironbow palette.
 In this image, my nose shows up green, so reading off the colour scale on the side, we can tell that the skin’s temperature was about 29 degrees Celsius. My skin here shows up red, so it was at a temperature of about 33 degrees Celsius. The exact temperature at the cross hairs is displayed here. I’m not sure why my nose skin was so cold. These faces show a far more even temperature.
In this image, my nose shows up green, so reading off the colour scale on the side, we can tell that the skin’s temperature was about 29 degrees Celsius. My skin here shows up red, so it was at a temperature of about 33 degrees Celsius. The exact temperature at the cross hairs is displayed here. I’m not sure why my nose skin was so cold. These faces show a far more even temperature.
Thermal imaging or Thermography, as it’s often called, has many, many applications. It’s used to monitor babies in humidicribs and it’s used in industry to detect blockages in pipes. This pipe contains warm water, but something that shouldn’t be there acts as an insulator, so that part of the pipe has a different temperature. There’s the blockage which in a factory, say, would have to be cleared. Water leakage in buildings also shows up clearly. Earlier we injected a small amount of water into a hole in the sheet of plaster. Infections or inflammations can also be assessed.
 The infrared camera filming me now works a little differently to a thermal imaging camera. This camera actually has an in-built, infrared light source. It produces infrared light with a frequency just below the frequency of visible red light. Electromagnetic waves in this region are often called “near infrared”, since they’re near the frequency of visible red light. The Mid and Far infrared regions are further from the visible spectrum. The infrared emitter is located here.
The infrared camera filming me now works a little differently to a thermal imaging camera. This camera actually has an in-built, infrared light source. It produces infrared light with a frequency just below the frequency of visible red light. Electromagnetic waves in this region are often called “near infrared”, since they’re near the frequency of visible red light. The Mid and Far infrared regions are further from the visible spectrum. The infrared emitter is located here.
You can see the infrared light coming from the camera if I hold up a mirror to the camera. The infrared light shines onto me and then reflects back into the camera. I can’t see it, but the camera’s sensor can. Unlike thermal imaging cameras, these kinds of infrared cameras produce realistic vision. Of course to produce an image, you need a lens. Near-Infrared light refracts when it hits glass, just like visible light does. Television remote controls send coded signals to the television using an infrared frequency that’s really close to the frequency of red light, 940nanometres in fact. This is so close to the visible spectrum that even though we can’t see it, most ordinary cameras can actually detect it.


 As we’ve seen, as things get hotter, they produce more and more infrared light, and the frequency of the infrared gets higher and higher. Using an infrared-detecting camera and a visible-light camera at the same time, we can see that the metal bar here emits lots of infrared when it’s heated. Eventually, when a high enough temperature is reached, typically about 500 degrees Celsius, the metal starts producing frequencies in the visible light region as well (which start at a frequency of 430THz). It’s still producing infrared light, but because it’s emitting visible red light as well, it is said to be red-hot.
As we’ve seen, as things get hotter, they produce more and more infrared light, and the frequency of the infrared gets higher and higher. Using an infrared-detecting camera and a visible-light camera at the same time, we can see that the metal bar here emits lots of infrared when it’s heated. Eventually, when a high enough temperature is reached, typically about 500 degrees Celsius, the metal starts producing frequencies in the visible light region as well (which start at a frequency of 430THz). It’s still producing infrared light, but because it’s emitting visible red light as well, it is said to be red-hot.
 A graph of the emitted infrared and visible light might look something like this. Notice here how it’s also producing light in the visible spectrum as well. At higher temperatures it gives off the other frequencies of visible light as well, so it said to be white hot.
A graph of the emitted infrared and visible light might look something like this. Notice here how it’s also producing light in the visible spectrum as well. At higher temperatures it gives off the other frequencies of visible light as well, so it said to be white hot.
The production of visible light from a really hot object is called incandescence. Incandescent light globes operate at temperatures of between 2000 to 3000 degrees Celsius. This one is still producing mostly infrared light, but because it’s now producing all the wavelengths of visible light as well, it appears white. So let’s now leave infrared light behind and look into the bright world of visible light.
Part F: Visible Light
Visible light occupies the very narrow region of the electromagnetic spectrum between 430THz and 790THz. Different frequencies and wavelengths are perceived as different colours.
 White light is made of a mixture of all the colours of the visible spectrum. The sun produces all of the wavelengths of visible light in equal measure so it appears white. White objects appear white because they reflect all the wavelengths of the visible spectrum. Red objects reflect red light but absorb the other colours. Blue objects reflect blue light and absorb the other colours.
White light is made of a mixture of all the colours of the visible spectrum. The sun produces all of the wavelengths of visible light in equal measure so it appears white. White objects appear white because they reflect all the wavelengths of the visible spectrum. Red objects reflect red light but absorb the other colours. Blue objects reflect blue light and absorb the other colours.
![]()
![]()
![]() Most light sources, like the sun, produce a range of wavelengths.
Most light sources, like the sun, produce a range of wavelengths.

Lasers, though, produce a single wavelength of light. This one is marked 633nm. It appears red. Lasers also produce waves which are all “in phase”; all the crests and the troughs line up exactly.
 Now we might call it visible light, but we only actually see it when it enters our eyes. There could be a 50 million watt laser beam shining right past me, but if it’s travelling in that direction, I’m not going to see it. Luckily it’s only a 1milliwatt laser. The light is now hitting my hand and reflecting off in every direction. Some is entering my eyes so I can see it and some is entering the camera, but we can’t see it along here.
Now we might call it visible light, but we only actually see it when it enters our eyes. There could be a 50 million watt laser beam shining right past me, but if it’s travelling in that direction, I’m not going to see it. Luckily it’s only a 1milliwatt laser. The light is now hitting my hand and reflecting off in every direction. Some is entering my eyes so I can see it and some is entering the camera, but we can’t see it along here.
 Movies often show laser lights as visible beams, but special effects are used to create these beams. If we could see the beams, we would also see them reflecting off the polished floor, here and here for example, but the special effects crew obviously didn’t draw them in.
Movies often show laser lights as visible beams, but special effects are used to create these beams. If we could see the beams, we would also see them reflecting off the polished floor, here and here for example, but the special effects crew obviously didn’t draw them in.
(If the cat burglar depicted in the movie Ocean’s Twelve did block the beam, how would a sensor know? A typical sensor in, say, a shop has a beam of light shining into it. If the beam is blocked, the sensor detects the drop in light intensity hitting it, and it makes a buzzing sound, or sets off an alarm. A moving laser beam doesn’t really make much sense. At one point, the beam is blocked by the statue. How does the alarm know the difference between a burglar and the statue? Once again, it doesn’t really make sense, but that’s not to say that Ocean’s Twelve wasn’t a good movie!)
 Smoke is often used at concerts because as a light beam moves through the smoke, some of the light reflects off the tiny smoke particles into our eyes and this allows us to see the light beam. The sun’s rays are sometimes visible from side on when they shine through mist.
Smoke is often used at concerts because as a light beam moves through the smoke, some of the light reflects off the tiny smoke particles into our eyes and this allows us to see the light beam. The sun’s rays are sometimes visible from side on when they shine through mist.

 Which leads us to the question of why the sky is blue. Most of the sunlight that hits our atmosphere passes straight through the atmosphere, because air is transparent.However, just as smoke particles can reflect and scatter a small amount of light, air can the same thing, but to a much, much, smaller extent. Though all of the wavelengths scatter a little, blue light happens to scatter slightly more than the rest.
Which leads us to the question of why the sky is blue. Most of the sunlight that hits our atmosphere passes straight through the atmosphere, because air is transparent.However, just as smoke particles can reflect and scatter a small amount of light, air can the same thing, but to a much, much, smaller extent. Though all of the wavelengths scatter a little, blue light happens to scatter slightly more than the rest.
A beam of sunlight shining directly onto you still appears white, since the amount of scattering is low, but all the other beams shining at an angle to you, appear slightly blue, because of the tiny amounts of blue light that scatter towards you. This scattered blue light coming from the trillions of light beams hitting the air is what makes the sky blue.
On the moon, there’s no air, so there’s no scattering. The sky, if you can call it that, looks black.
When the sun is low on the horizon, the light has to travel through more of the atmosphere, so a lot more light is scattered, especially when there’s lots of dust in the air. The sky often turns red and the sun appears to change colour too.



