The Shedding Light on Atoms series gives students the perfect introduction to the world of atoms. Using amazing demonstrations and animations we take students on a journey of discovery to explain not just what we know about atoms, but also how we know what we know about atoms!
In Episode 6, Electron Shells, we look at how electrons are arranged within atoms in electron shells, and at how the Periodic Table is organised to reflect these arrangements. We demonstrate a variety of experiments which led to the discovery of electron shells and finish by taking a brief look at the size of atoms.
The preview video below contains a 4-minute excerpt followed by a 1-minute trailer.
 The Episode 6 Question Sheet for Students:
The Episode 6 Question Sheet for Students:
![]() QS6 Electron Shells.
QS6 Electron Shells. ![]()
![]() If you have ClickView, watch the whole episode here.
If you have ClickView, watch the whole episode here.
![]() If you have Learn360, watch the whole episode here.
If you have Learn360, watch the whole episode here.
![]() If you have Films on Demand, watch the whole episode here.
If you have Films on Demand, watch the whole episode here.
![]() If you have Classroom Video, watch the whole episode here.
If you have Classroom Video, watch the whole episode here.
![]() Most of our videos are also available on SAFARI Montage. Just log in and do a quick search.
Most of our videos are also available on SAFARI Montage. Just log in and do a quick search.
![]() Don’t have any of the above? Rent or buy the Shedding Light series and/or individual programs from our Vimeo page!!
Don’t have any of the above? Rent or buy the Shedding Light series and/or individual programs from our Vimeo page!!
 Here’s an A4 printable Periodic Table that that includes notes that will help students learn about isotopes, electron shell configurations, ions, and much more.
Here’s an A4 printable Periodic Table that that includes notes that will help students learn about isotopes, electron shell configurations, ions, and much more.
The pdf version.
The Word (docx) version.
The Transcript (which can be used as a textbook)
Contents:
Part A: Introduction: Atoms are made of a nucleus of protons and neutrons which is surrounded by orbiting electrons. The electrons occupy only certain fixed energy levels called electron shells.
Part B: The Atom Sheds Light: The discovery of electrons shells can be traced back to the discovery that elements which are in a hot gaseous state glow with a specific range of colours which is unique to each element. We demonstrate the use of flame tests and spectroscopes and explain how they were used to discover new elements.
Part C: The Discovery of Electron Shells: By analysing the light emitted by atoms and by firing X-rays at various crystals, scientists in the 1910s discovered that electrons don’t orbit randomly around the nucleus (like planets orbit the sun), but orbit only in electron shells with a specific range of energies.
Part D: Electron Configurations: Electrons occupy electron shells. Each shell that surrounds the nucleus can hold only a certain number of electrons. The Periodic Table organises the elements into Groups and Periods (columns and rows) based largely on how the electrons are arranged in each element’s electron shells.
Part E: The Size of Atoms: The size of an atom is determined by how spread out the electrons are. We demonstrate using a solid steel sphere how small atoms actually are.
Part A: Introduction
Atoms are the tiny tiny building blocks that make up everything.  Elements, like Lithium, Actinium and Osmium, just to name a random selection, are substances made up of only one type of atom. Compounds, like water, salt, and literally millions of other examples, are made of two or more types of atoms that chemically bond together, although we haven’t yet covered how this actually happens.
Elements, like Lithium, Actinium and Osmium, just to name a random selection, are substances made up of only one type of atom. Compounds, like water, salt, and literally millions of other examples, are made of two or more types of atoms that chemically bond together, although we haven’t yet covered how this actually happens.
Though for a long time it was thought that atoms were the smallest units of all matter, scientists in the late 1800s and early 1900s discovered that atoms were actually made up of even smaller particles,  which were named protons, neutrons and electrons. In fact, all of the different types of atoms are made of just these three particles. The protons and neutrons make up the nucleus of the atom and the electrons spin around the nucleus at enormous speeds effectively making the atom more or less spherical.
which were named protons, neutrons and electrons. In fact, all of the different types of atoms are made of just these three particles. The protons and neutrons make up the nucleus of the atom and the electrons spin around the nucleus at enormous speeds effectively making the atom more or less spherical.
The number of protons in the nucleus of an atom defines the type of atom that it is. Every Hydrogen atom has 1 proton in it nucleus, every Helium atom has 2 protons in it nucleus, every Lithium atom three and so on. Most hydrogen atoms have no neutrons, so the one and only proton is the nucleus, most Helium atoms have 2 neutrons, and most Lithium atoms have 4 neutrons, but the neutron number can vary, within limits, and it doesn’t really affect the way an atom will chemically react. Atoms have an equal number of electrons and protons. The number of protons an atom has is called the atomic number. It can be seen on most Periodic Tables.
 Soon after the basic structure of the atom was discovered, various experiments and a mathematical analysis of all the data showed that electrons can occupy only certain fixed energy levels that are called electron shells, which are usually represented simply as circles, although remember that atoms are 3D of course.
Soon after the basic structure of the atom was discovered, various experiments and a mathematical analysis of all the data showed that electrons can occupy only certain fixed energy levels that are called electron shells, which are usually represented simply as circles, although remember that atoms are 3D of course.
The discovery of electron shells helped explain lots of things about the way that atoms behaved, especially about the way they joined together to form compounds, and so, it’s electron shells that we’ll be looking at in this episode.
Part B: The Atom Sheds Light
 It had been known for a long time that when different gases are heated, they produce different colours. These powders all look very much alike, except of course for the green one. We have copper chloride, lithium chloride, potassium chloride and so on. They’re all types of what scientists call salts. When we use the word salt we usually mean the salt that we eat: table salt, or sodium chloride, NaCl. However, there are lots of different types of salts, some of which we have here. Now though they look similar we can use what’s called a flame test to tell them apart.
It had been known for a long time that when different gases are heated, they produce different colours. These powders all look very much alike, except of course for the green one. We have copper chloride, lithium chloride, potassium chloride and so on. They’re all types of what scientists call salts. When we use the word salt we usually mean the salt that we eat: table salt, or sodium chloride, NaCl. However, there are lots of different types of salts, some of which we have here. Now though they look similar we can use what’s called a flame test to tell them apart.

 If we take some copper chloride and heat it, a small amount is vaporised and it glows with a distinctive blue-green light. Lithium chloride vapour produces bright red light… potassium chloride orange light with a hint of light purple… sodium chloride, bright yellow light… barium chloride pale yellow light… and calcium chloride red, orange and yellow light…
If we take some copper chloride and heat it, a small amount is vaporised and it glows with a distinctive blue-green light. Lithium chloride vapour produces bright red light… potassium chloride orange light with a hint of light purple… sodium chloride, bright yellow light… barium chloride pale yellow light… and calcium chloride red, orange and yellow light…
By the way, the many different colours of fireworks are produced by various salts which are mixed into each firework’s flammable powders. Fireworks are basically giant flame tests in the sky.
Now though some colours produced in a flame test are obviously quite distinct, many are fairly similar.
 In 1814, German scientist Joseph von Fraunhofer, invented the first spectroscope, an instrument that makes it possible to determine the exact colours produced in flam tests. If we look at the coloured flame of, for example, calcium chloride vapour through a spectroscope, well, here we’re actually filming it, we can see, as vertical lines, the individual colours of light produced in the flame.
In 1814, German scientist Joseph von Fraunhofer, invented the first spectroscope, an instrument that makes it possible to determine the exact colours produced in flam tests. If we look at the coloured flame of, for example, calcium chloride vapour through a spectroscope, well, here we’re actually filming it, we can see, as vertical lines, the individual colours of light produced in the flame.  The spectroscope breaks the light up into its component colours with each line being a specific wavelength of light. These numbers indicate the wavelength of the light. This light, for example, has a wavelength of just over 550 nanometres.
The spectroscope breaks the light up into its component colours with each line being a specific wavelength of light. These numbers indicate the wavelength of the light. This light, for example, has a wavelength of just over 550 nanometres.
 So, how does a spectroscope work? Well, really hot objects like the sun and incandescent light bulbs, when they’re not broken, produce white light, which is actually a mixture of all the colours of the rainbow. When a white light beam shines from say air into glass at an angle it refracts, that is, it changes direction.
So, how does a spectroscope work? Well, really hot objects like the sun and incandescent light bulbs, when they’re not broken, produce white light, which is actually a mixture of all the colours of the rainbow. When a white light beam shines from say air into glass at an angle it refracts, that is, it changes direction.
However, all the component colours of white light refract by a slightly different amount, so if white light is shone through a prism where it refracts twice, all the colours (that is, all the different wavelengths of the light) spread out quite a lot and we can see the colours of the so-called visible spectrum. We often simply call this continuous visible spectrum a rainbow.
Basically, all hot solids, liquids and high-pressure gases produce a continuous spectrum (although the light isn’t necessarily white).
 However, in a flame test, the light being produced by the low-pressure hot gases is not white and it’s not a continuous spectrum, it’s made up of only certain wavelengths. When this light hits the prism, it spreads out and the individual wavelengths produced in the flame can be seen and studied.
However, in a flame test, the light being produced by the low-pressure hot gases is not white and it’s not a continuous spectrum, it’s made up of only certain wavelengths. When this light hits the prism, it spreads out and the individual wavelengths produced in the flame can be seen and studied.
The set of wavelengths that an element emits when it is in the form of a low-pressure hot gas is called its emission spectrum.
 Sodium chloride vapour produces two specific wavelengths of yellow light, it’s a little hard to see but there are two, while the glow of strontium chloride is made up of different wavelengths again. A spectroscope makes it fairly easy to tell elements apart. This image shows von Fraunhofer demonstrating his spectroscope in about 1820.
Sodium chloride vapour produces two specific wavelengths of yellow light, it’s a little hard to see but there are two, while the glow of strontium chloride is made up of different wavelengths again. A spectroscope makes it fairly easy to tell elements apart. This image shows von Fraunhofer demonstrating his spectroscope in about 1820.
 In the 1860s, German scientists Gustav Kirchhoff and Robert Bunsen, the inventor of the famous Bunsen burner, discovered that each element produces a unique set of wavelengths. They built a really accurate spectrometer and analysed the emission spectrums of lots of elements. You can see the prism they used here. When flame testing various minerals in the mineral water
In the 1860s, German scientists Gustav Kirchhoff and Robert Bunsen, the inventor of the famous Bunsen burner, discovered that each element produces a unique set of wavelengths. They built a really accurate spectrometer and analysed the emission spectrums of lots of elements. You can see the prism they used here. When flame testing various minerals in the mineral water  from a place called Durkheim in Germany, they found certain blue spectral lines that didn’t correspond to any known element. They had in fact discovered a new element which they called caesium (or cesium) after the Latin word “caesius” which means light blue. A pure sample of caesium wasn’t obtained however for another 20 years or so.
from a place called Durkheim in Germany, they found certain blue spectral lines that didn’t correspond to any known element. They had in fact discovered a new element which they called caesium (or cesium) after the Latin word “caesius” which means light blue. A pure sample of caesium wasn’t obtained however for another 20 years or so.
Bunsen and Kirchhoff also discovered Rubidium from its emission spectrum, in a mineral called lepidolite. Certain deep red spectral lines had never been seen before in a flame test, so the new element was named rubidium after the Latin word rubeus which means deep red. (Rubidium has nothing to do with the ruby, except that both words come from the same original Latin word.)
So Rubidium and Caesium are named after specific colours in their emission spectrums which no other element emits in a flame test. They could just as easily have been called deepredium and lightbluium.
Many other elements were also later discovered by analysing emission spectrums, although purifying the elements needed various chemical techniques, like, for example, electrolysis, which we looked at in Episode 4 of our series.
 In the late 1800s and early 1900s, scientists started using these so-called discharge tubes to study emission spectrums. Really accurate emission spectrums could be obtained from individual elements. Inside this metal case is a glass tube with pure neon gas in it. A small electric current is made to flow through the neon gas and the gas gives off a warm reddish glow. The spectroscope reveals the unique set of wavelengths which are produced by pure Neon gas.
In the late 1800s and early 1900s, scientists started using these so-called discharge tubes to study emission spectrums. Really accurate emission spectrums could be obtained from individual elements. Inside this metal case is a glass tube with pure neon gas in it. A small electric current is made to flow through the neon gas and the gas gives off a warm reddish glow. The spectroscope reveals the unique set of wavelengths which are produced by pure Neon gas.
 This tube contains a small amount of mercury. When it’s heated and a current passes through it, the mercury turns into a gas and glows with a blue-green light, which the spectroscope shows is made of just a few specific wavelengths. The pure sodium vapour in this discharge tube glows with two specific, almost identical, wavelengths of yellow light.
This tube contains a small amount of mercury. When it’s heated and a current passes through it, the mercury turns into a gas and glows with a blue-green light, which the spectroscope shows is made of just a few specific wavelengths. The pure sodium vapour in this discharge tube glows with two specific, almost identical, wavelengths of yellow light.
Simple discharge tubes like these ones led to the development of really efficient neon lights and fluorescent tubes which transformed the way cities and buildings were lit.

Really accurate and detailed emission spectrums of elements are now fairly easy to produce and to photograph, and they’re used, for example, to analyse what atoms are found within different minerals. By the way the plural of the word spectrum is either spectrums or spectra.
| Most of the images of elements used in the Shedding Light on Atoms series were taken by Heinrich Pniok. Visit http://pse-mendelejew.de/en/ to see his collection. |
So, what was causing all these different atoms to produce all these specific wavelengths of light. Well, it came down to how the electrons were arranged within each atom.
Part C: The Discovery of Electron Shells
 We saw in our last episode that in 1897, JJ Thomson discovered electrons by applying a really high voltage between two metal plates. We also saw how, about a decade later, Ernest Rutherford fired alpha particles at an extremely thin piece of gold foil and found that though most alpha particles passed straight through the gold foil, a small number were deflected by large amounts, which led him to the conclusion that atoms were made of a small positively charged nucleus which was surrounded by negatively charged fast-moving electrons.
We saw in our last episode that in 1897, JJ Thomson discovered electrons by applying a really high voltage between two metal plates. We also saw how, about a decade later, Ernest Rutherford fired alpha particles at an extremely thin piece of gold foil and found that though most alpha particles passed straight through the gold foil, a small number were deflected by large amounts, which led him to the conclusion that atoms were made of a small positively charged nucleus which was surrounded by negatively charged fast-moving electrons.
 In 1913, Danish scientist Niels Bohr improved upon Rutherford’s model of the atom by suggesting that the electrons don’t just orbit in random positions around the nucleus but move only in certain fixed energy levels which we now usually call electron shells. He (Bohr) used these fixed energy levels to explain why elements give off only certain wavelengths of light when either undergoing a flame test or when an electric current is passed through them in a discharge tube.
In 1913, Danish scientist Niels Bohr improved upon Rutherford’s model of the atom by suggesting that the electrons don’t just orbit in random positions around the nucleus but move only in certain fixed energy levels which we now usually call electron shells. He (Bohr) used these fixed energy levels to explain why elements give off only certain wavelengths of light when either undergoing a flame test or when an electric current is passed through them in a discharge tube.
| When an electron is in a higher shell than normal, the atom is said to be in an “excited” state. |
 So, let’s look at what happens in a sodium vapour discharge tube. This box contains an electrical device which generates a really really high voltage between two metal plates inside the tube. Normally, electricity, which is basically the movement of electrons in a circuit, needs a conductor to flow through, like a metal wire. However, if the voltage is high enough, electrons are forced off the cathode and they stream towards the anode, even if there is nothing in between the two.
So, let’s look at what happens in a sodium vapour discharge tube. This box contains an electrical device which generates a really really high voltage between two metal plates inside the tube. Normally, electricity, which is basically the movement of electrons in a circuit, needs a conductor to flow through, like a metal wire. However, if the voltage is high enough, electrons are forced off the cathode and they stream towards the anode, even if there is nothing in between the two.
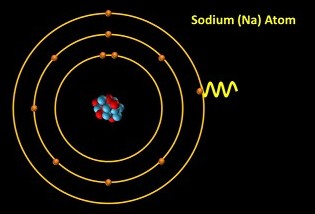 Bohr suggested that an electron streaming through the sodium gas occasionally slams into a sodium atom’s electron and sends that electron into a higher electron shell, which has a higher energy level. Remember, the electrons are constantly moving but we’ve shown them stationary.
Bohr suggested that an electron streaming through the sodium gas occasionally slams into a sodium atom’s electron and sends that electron into a higher electron shell, which has a higher energy level. Remember, the electrons are constantly moving but we’ve shown them stationary.
Let’s watch it again. The free electron crashes into the sodium atom’s electron and gives it extra energy, sending it into a higher energy level. However, the electron can’t stay there for very long and so it drops back down to its normal electron shell almost immediately. But what happens to the extra energy that it had? Well, let’s rewind for a moment. In fact, when the electron drops back down, the energy is given off in the form of light energy whose wavelength (and therefore colour) is related to the energy difference between the two electron shells.
Of course, electrons can jump up 2 or more energy levels, and when they drop back down either directly or from shell to shell, they release light with a range of different wavelengths. Emission spectrums, Bohr’s theory suggested, were caused by the movement of electrons between electron shells.
So, even though we can’t see atoms, the light that they emit tells us a lot about them.
 Now remember, the electron shells are NOT actual spheres, or solid objects or anything. They’re best thought of either as the regions that the electrons move around in really really fast, or as energy levels. The orange circles that we’ve been drawing that surround the nucleus aren’t really there either; they just help us visualize the atom.
Now remember, the electron shells are NOT actual spheres, or solid objects or anything. They’re best thought of either as the regions that the electrons move around in really really fast, or as energy levels. The orange circles that we’ve been drawing that surround the nucleus aren’t really there either; they just help us visualize the atom.
Bohr had to use a lot of complicated mathematics and physics to properly express what was going on, and even then, the equations could only explain the emission of hydrogen atoms, which had only one electron. The emission spectra of the other atoms didn’t fit very well at all with Bohr’s equations. Never-the-less, his ideas were brilliant, and, after a little bit of refinement, they opened up a whole new area of science called quantum mechanics.
Because each atom has a different number of protons and electrons, the electron shells all have different energy levels, so each atom’s emission spectrum is unique.
In our last episode, we saw how the study of radioactivity helped scientists work out the structure of the atom, and vice versa. Well, the study of light also helped us work out the structure of the atom, and vice versa! So how many electrons are in each shell?
 In 1912, Australian scientist William Lawrence Bragg, found that, by bouncing X-rays off tiny crystals and recording the patterns produced, he could determine how the atoms were arranged within the crystals. I can demonstrate the patterns with a laser and a slide which has a series of very very narrow slits in it. The slits, which are actually double slits can be seen in this close up… here and here for example. The fact that this bar is 1cm wide gives you an indication of how narrow they are.
In 1912, Australian scientist William Lawrence Bragg, found that, by bouncing X-rays off tiny crystals and recording the patterns produced, he could determine how the atoms were arranged within the crystals. I can demonstrate the patterns with a laser and a slide which has a series of very very narrow slits in it. The slits, which are actually double slits can be seen in this close up… here and here for example. The fact that this bar is 1cm wide gives you an indication of how narrow they are.

 Normally, the laser beam is very tight. However, when the light beam passes through the tiny slits in the slide, it spreads out and forms patterns on the screen. The patterns change depending on the width of the slits and how apart they are.
Normally, the laser beam is very tight. However, when the light beam passes through the tiny slits in the slide, it spreads out and forms patterns on the screen. The patterns change depending on the width of the slits and how apart they are.
 Now similar kinds of patterns occur when X-rays pass between the atoms in a crystal. It was very complicated stuff, but, by analysing these patterns, Bragg, and other scientists, were eventually able to work out not just how the atoms were arranged in various crystals, but also, by around the late 1920s, how many electron shells each atom had and how many electrons were in each shell. This led directly to an understanding of how atoms bond together to form compounds. So let’s have a look at the numbers.
Now similar kinds of patterns occur when X-rays pass between the atoms in a crystal. It was very complicated stuff, but, by analysing these patterns, Bragg, and other scientists, were eventually able to work out not just how the atoms were arranged in various crystals, but also, by around the late 1920s, how many electron shells each atom had and how many electrons were in each shell. This led directly to an understanding of how atoms bond together to form compounds. So let’s have a look at the numbers.
Part D: Electron Configurations
 The innermost electron shell of an atom is called the first shell. Since it is so small, it can hold a maximum of only two electrons. If any more tried to cram in, they would be repelled by the electrons that are already there. The next shell is called the second shell and, being larger, can hold a maximum of 8 electrons. Next comes the third shell which can hold a maximum of 18 electrons.
The innermost electron shell of an atom is called the first shell. Since it is so small, it can hold a maximum of only two electrons. If any more tried to cram in, they would be repelled by the electrons that are already there. The next shell is called the second shell and, being larger, can hold a maximum of 8 electrons. Next comes the third shell which can hold a maximum of 18 electrons.
![]() Silicon atoms have 2 electrons in their first shell, 8 in their second and 4 in their third. To express this, we say that its electron configuration is 2, 8, 4.
Silicon atoms have 2 electrons in their first shell, 8 in their second and 4 in their third. To express this, we say that its electron configuration is 2, 8, 4.
Let’s look at the electron configurations of the first 18 elements, beginning of course with the elements in Period 1. Remember, the horizontal rows of the Periodic Table are called Periods.
 A hydrogen atom has only one shell which has only 1 electron in it. Its electron configuration is simply 1. A helium atom also has only one shell, but there are two electrons in it. Its electron configuration is 2, and the shell is now full. Let’s now move onto Period 2. Lithium atoms have 3 electrons. Two of them fit into the first shell, but a second shell is needed for the third electron. Lithium’s electron configuration is 2, 1. Now I’ll just point out that none of these atoms are drawn to scale. Beryllium comes next. It has 4 electrons and an electron configuration of 2, 2. Boron has an electron configuration of 2, 3, carbon 2, 4, nitrogen 2, 5, oxygen 2, 6, fluorine 2, 7 and neon, 2, 8. Both of Neon’s electron shells are full.
A hydrogen atom has only one shell which has only 1 electron in it. Its electron configuration is simply 1. A helium atom also has only one shell, but there are two electrons in it. Its electron configuration is 2, and the shell is now full. Let’s now move onto Period 2. Lithium atoms have 3 electrons. Two of them fit into the first shell, but a second shell is needed for the third electron. Lithium’s electron configuration is 2, 1. Now I’ll just point out that none of these atoms are drawn to scale. Beryllium comes next. It has 4 electrons and an electron configuration of 2, 2. Boron has an electron configuration of 2, 3, carbon 2, 4, nitrogen 2, 5, oxygen 2, 6, fluorine 2, 7 and neon, 2, 8. Both of Neon’s electron shells are full.
Let’s now move on to Period 3 of the Periodic Table.
Sodium, needs three shells to fit all of its 11 electrons. Its electron configuration is 2, 8, 1. Magnesium has 12 electrons and an electron configuration of 2, 8, 2. We then have Aluminium, or aluminum if you prefer with 2, 8, 3, Silicon with 2, 8, 4, Phosphorus with 2, 8, 5, Sulfur with 2, 8, 6, Chlorine with 2, 8, 7 and finally, Argon with 2, 8, 8.
 Notice how the two elements in Period 1 have only 1 electron shell, the elements in Period 2 have 2 electron shells, and the elements in Period 3 have three electron shells. This pattern, in fact, applies to all the periods. The Period that an element is in on the Periodic Table tells you the number of electron shells that the element has. All of the elements in Period 4 have four electron shells, all of the elements in Period 5 have 5 electron shells and so on.
Notice how the two elements in Period 1 have only 1 electron shell, the elements in Period 2 have 2 electron shells, and the elements in Period 3 have three electron shells. This pattern, in fact, applies to all the periods. The Period that an element is in on the Periodic Table tells you the number of electron shells that the element has. All of the elements in Period 4 have four electron shells, all of the elements in Period 5 have 5 electron shells and so on.
 Now probably more importantly, there’s another pattern involving the Periodic Table and electron shells and it has to do with the arrangements of the elements into Groups, that is, into the Table’s vertical columns. Notice how the atoms in Group 1, hydrogen, lithium and sodium all have 1 electron in their outer shell.
Now probably more importantly, there’s another pattern involving the Periodic Table and electron shells and it has to do with the arrangements of the elements into Groups, that is, into the Table’s vertical columns. Notice how the atoms in Group 1, hydrogen, lithium and sodium all have 1 electron in their outer shell.
 This in fact applies to all the elements in group 1. They all have 1 outer-shell electron. The atoms in Group 2, beryllium and magnesium, and all the rest, have 2 electrons in their outer shell.
This in fact applies to all the elements in group 1. They all have 1 outer-shell electron. The atoms in Group 2, beryllium and magnesium, and all the rest, have 2 electrons in their outer shell.
To continue the trend, we have to skip over to Group 13, not Group 3, but Group 13. In Group 13, the atoms all have 3 outer shell electrons.
The atoms in Group 14 all have 4 outer-shell electrons, and the pattern continues until Group 18, where all the atoms have 8 outer-shell electrons.
Helium is the only odd one out. It’s placed into Group 18 because it’s a very unreactive gas, just like all the other gases in Group 18.
The number of electrons in the outer shell of an atom is really important because it plays a major part in the way that the atom chemically bonds to other atoms, but we’ll talk more about this later.
The atoms that make up the elements in Groups 3 to 12 usually have either 1 or 2 electrons in their outer shell. These elements, which are all metals, are often called Transition Metals or Transition Elements.
 So, by looking at the position on any element on the periodic table we can get a lot of information about it. If we look at, say, iodine, we can tell that, because it’s in Group 17, it has 7 electrons in its outer shell, and because it’s in Period 5, we know it has five electron shells. Barium, being in Group 2 has 2 electrons in its outer shell and being in Period 6 has six electron shells. Nickel, being in Period 4 has four electron shells, but because it’s a transition metal, we can’t tell from the periodic table how many electrons it has in its outer shell. It happens to have 2, though. (Its electron configuration is 2, 8, 16, 2.)
So, by looking at the position on any element on the periodic table we can get a lot of information about it. If we look at, say, iodine, we can tell that, because it’s in Group 17, it has 7 electrons in its outer shell, and because it’s in Period 5, we know it has five electron shells. Barium, being in Group 2 has 2 electrons in its outer shell and being in Period 6 has six electron shells. Nickel, being in Period 4 has four electron shells, but because it’s a transition metal, we can’t tell from the periodic table how many electrons it has in its outer shell. It happens to have 2, though. (Its electron configuration is 2, 8, 16, 2.)
Let’s now take a quick look at the electron configurations of all the elements in Period 4, which don’t quite follow the same pattern that we’ve already seen.
 Since the electron configuration of Argon, the last Period 3 element is 2, 8, 8, and the third shell can actually hold a maximum of 18 electrons, you might think that the electron configuration of Potassium, which comes after Argon, should be 2, 8, 9, but it isn’t. It is in fact 2, 8, 8, 1, and calcium’s is 2, 8, 8, 2. The fourth shell starts filling up before the third shell is complete. It’s just one of those things in nature. The third shell actually continues to fill in the Transition metals. Scandium’s electron configuration is 2, 8, 9, 2, titanium’s is 2, 8, 10, 2, and the pattern continues, although not perfectly, until we get to zinc, which has an electron configuration of 2, 8, 18, 2. The third shell is now full, so what is gallium’s electron configuration? If you said 2, 8, 18, 3, you are correct! Gallium has three outer-shell electrons just like all the other elements in Group 13. The pattern then continues as it did in the previous periods: 2, 8, 18, 4; 2, 8, 18, 5 and so on until we get to Krypton (2, 8, 18, 8).
Since the electron configuration of Argon, the last Period 3 element is 2, 8, 8, and the third shell can actually hold a maximum of 18 electrons, you might think that the electron configuration of Potassium, which comes after Argon, should be 2, 8, 9, but it isn’t. It is in fact 2, 8, 8, 1, and calcium’s is 2, 8, 8, 2. The fourth shell starts filling up before the third shell is complete. It’s just one of those things in nature. The third shell actually continues to fill in the Transition metals. Scandium’s electron configuration is 2, 8, 9, 2, titanium’s is 2, 8, 10, 2, and the pattern continues, although not perfectly, until we get to zinc, which has an electron configuration of 2, 8, 18, 2. The third shell is now full, so what is gallium’s electron configuration? If you said 2, 8, 18, 3, you are correct! Gallium has three outer-shell electrons just like all the other elements in Group 13. The pattern then continues as it did in the previous periods: 2, 8, 18, 4; 2, 8, 18, 5 and so on until we get to Krypton (2, 8, 18, 8).
The electron configuration patterns in Groups 4 and above are a little complicated, but I just thought I’d show you anyway so that you can see why the Periodic Table is arranged the way it is. At this stage, knowing the electron configurations of just the first 20 elements is probably enough.
Because of the way electrons fill electron shells, no element is made of atoms that have more than eight electrons in their outer shell, except for Palladium, whose atoms have an electron configuration of 2, 8, 18, 18. Once again it’s just one of those things.
When Mendeleev first developed the Periodic Table, he labelled the Groups from 1 to 8, placing what are mostly transition metals into Group 8. As more and more discoveries were made, the Periodic Table went through various modifications.
In the 1920s, when scientists figured out the patterns in the way that electrons fill electron shells, the transition metals started being placed in between Groups 2 and 3 (like they are in this 1925 Periodic Table). In 1990, the Groups were renumbered so that they went consecutively from 1 to 18. (The transition elements now make up Groups 3 to 12, and what was Group 3 is now Group 13, what was Group 4 is now Group 14 and so on. There’s a little more detail in Bonus Feature 1 of this program.)
 Now in case you’re wondering, the so-called lanthanides and actinides, are placed at the bottom of the Periodic Table simply to allow the table to fit more easily onto a normal screen. In reality they belong in these spaces here, so the Periodic Table should really be this wide and it is sometimes drawn this way, but usually it’s just not practical because, being 32 columns wide, the text is too small.
Now in case you’re wondering, the so-called lanthanides and actinides, are placed at the bottom of the Periodic Table simply to allow the table to fit more easily onto a normal screen. In reality they belong in these spaces here, so the Periodic Table should really be this wide and it is sometimes drawn this way, but usually it’s just not practical because, being 32 columns wide, the text is too small.
Part E: The Size of Atoms.
Even though most of the mass of the atom is concentrated in the atom’s nucleus, the size of the atom is determined by how spread out the electrons are and this can vary because, even though we’ve been showing the electrons moving in circular paths, they actually kind of wobble about in their orbits. As a result, there is no way to determine the exact size of an atom. But we can get good approximations!
I mentioned earlier that by analysing the patterns produced by X-rays passing between crystals, scientists were able to work out how the atoms were arranged in the crystals. Over time, they were eventually able to estimate the size of the atoms. It turns out that atoms are extremely small!

 This periodic table shows the approximate diameters of most atoms expressed in picometres which can also be pronounced as picometres. A picometer or picometre is an unimaginably small 1 trillionth of a metre, which is the same as 1 millionth millionth of a metre. It’s pretty much impossible to visualise, but iron atoms for example, have a diameter of about 280 picometres.
This periodic table shows the approximate diameters of most atoms expressed in picometres which can also be pronounced as picometres. A picometer or picometre is an unimaginably small 1 trillionth of a metre, which is the same as 1 millionth millionth of a metre. It’s pretty much impossible to visualise, but iron atoms for example, have a diameter of about 280 picometres.
 It’s very hard to picture 280 picometres, so let me illustrate with this solid steel sphere, which has a diameter of 6cm. Steel by the way is about 99% iron atoms.
It’s very hard to picture 280 picometres, so let me illustrate with this solid steel sphere, which has a diameter of 6cm. Steel by the way is about 99% iron atoms.
The size of the whole earth is to the size of this steel sphere what the size of the steel sphere is to the size of every iron atom that’s in it.
So, there are about as many iron atoms in this 6cm-diameter solid steel sphere as it would take to fill the entire earth with spheres of this size. That’s still very hard to imagine, so, as we zoom out using Google Earth, I’ll say it one more time. In size, the whole earth is to the steel sphere, what the steel sphere is to an iron atom.
 In fact the steel sphere is just over 200 million times wider than an iron atom and the whole earth is also just over 200 million times wider than the steel sphere. Incredible! I’m still having trouble comprehending it.
In fact the steel sphere is just over 200 million times wider than an iron atom and the whole earth is also just over 200 million times wider than the steel sphere. Incredible! I’m still having trouble comprehending it.
(You can go through the calculations at the end.)
There isn’t really a huge variation in the size of all the different types of atoms. Most of them are between about 200 and 400 picometres in diameter. There are, however, two fairly clear trends.
 Firstly, not including the transition metals, as you move down each group, atoms tend to get larger. For example, in Group 1, hydrogen atoms have a diameter of a little over 100 picometres, while the atoms below it get progressively bigger. They get bigger because, as you can see, they have more electron shells. The same sort of thing happens in, for example, Group 1.
Firstly, not including the transition metals, as you move down each group, atoms tend to get larger. For example, in Group 1, hydrogen atoms have a diameter of a little over 100 picometres, while the atoms below it get progressively bigger. They get bigger because, as you can see, they have more electron shells. The same sort of thing happens in, for example, Group 1.
 The second trend is that as you move across the periods from left to right, atoms tend to get smaller. For example, in Period 2, where all the atoms have two electron shells, the lithium atoms have a diameter of just under 300 picometres while the neon atoms have a diameter of only about 80 picometres. Atoms tend to get smaller as you move across a period because the number of protons in the nucleus of each atom increases. When there are more protons, they apply a greater force on the electrons, effectively pulling them into a smaller space.
The second trend is that as you move across the periods from left to right, atoms tend to get smaller. For example, in Period 2, where all the atoms have two electron shells, the lithium atoms have a diameter of just under 300 picometres while the neon atoms have a diameter of only about 80 picometres. Atoms tend to get smaller as you move across a period because the number of protons in the nucleus of each atom increases. When there are more protons, they apply a greater force on the electrons, effectively pulling them into a smaller space.
So, the largest atoms are at the bottom left of the Periodic Table, while the smallest atoms are at the top right.
Now in Episode 4 of our series, which we called “The Periodic Table”, we saw how Dmitri Mendeleev organised the elements into his Periodic Table mostly on the basis of how each element chemically reacted with oxygen, the atoms of all Group 1 elements for example react with oxygen atoms in a 2:1 ratio, and with hydrogen, you can see the 4:1, 3:1, 2:1 and 1:1 pattern as you move from left to right. In this Episode of our series, we’ve been looking at how the Periodic Table is arranged according to electron shells.
 So what does this tell us? It tells us that the way that electrons are arranged in electron shells plays a major role in how atoms bond together to form compounds and it dictates what combinations of atoms are possible.
So what does this tell us? It tells us that the way that electrons are arranged in electron shells plays a major role in how atoms bond together to form compounds and it dictates what combinations of atoms are possible.
So it’s probably not surprising that the discovery of electron shells soon led to an understanding of atomic bonding, which is what we’ll be looking at in our next episode. See you then.
Shedding Light on Atoms Episode 6: Electron Shells Question Sheet

Part A:
1. Atoms are made of three subatomic particles: ______________, _____________, and _______________.
2. ______________ and _____________ make up the nucleus of an atom while __________________ orbit around the nucleus.
3. The atomic number of boron, B, is 5 and so, by definition, every boron atom has _____ protons in its nucleus. Iron atoms have _____ protons in their nucleus and so they have an atomic number of _____.
Part B:
4. Lithium chloride, sodium chloride, and barium chloride are all different types of “salts”, and they all look very similar to each other. Describe an easy way of telling them apart.
5. An element’s emission spectrum is the set of wavelengths of light that it emits when it is in the form of a low-pressure hot gas. In what way is an emission spectrum like a human fingerprint?

 6. The emission spectrum on the left comes from __________________ while the emission spectrum on the right comes from __________________.
6. The emission spectrum on the left comes from __________________ while the emission spectrum on the right comes from __________________.
Part C:
7. In 1913, Danish scientist Neils Bohr suggested that electrons can occupy only certain energy levels which were named _________________ _______________.
8. According to Bohr’s theory, how is light produced by atoms in discharge tubes (and in flame tests)?
Part D:
9. Label sodium’s electron-shell diagram.

10. Fill in the table below.
11. What is the electron configuration of sodium (Na)?
12.Fill in the electron configurations of the elements in the table below. Carbon’s has already been done.

13. Draw electron configuration diagrams for hydrogen, carbon, and oxygen atoms.
14. The atoms of all (both) elements in Period 1 have ______ electron shell, the atoms of all elements in Period 2 have ______ electron shells, and the atoms of all elements in Period 3 have ______ electron shells.
15. What does the Period number (that an element is in) tell you?
16. The atoms of the elements in Groups 1 & 2 and 13-18 have a specific number of electrons in their outer shell. Fill in the rest of the table below. (The Group number is NOT necessarily the same as the number of electrons in an atom’s shell.)
17. All the atoms of the Group 18 elements have ______ electrons in their outer shell, except for helium atoms. Why is helium placed into Group 18?
18. The atoms of most transition metals (metals in Groups 3 to 12) have either ______ or ______ electrons in their outer shells.
19. Atoms in their pure, elemental form (that is, atoms which have not chemically reacted with other atoms) have a maximum of _______ electrons in their outer shells (except for __________________ atoms.)
20. Fill in the table below. You will need a periodic table.

21. Potassium, K, has an electron configuration of ______________ and Calcium, Ca, has an electron configuration of _________________.
Part E:
22. What is a picometre (pm)?
23. In picometres, what is the approximate diameter of an iron (Fe) atom?
24. Why can’t an exact diameter of a single atom be measured?
25. The earth has a diameter of 12, 742km. Exactly how many times wider is this than a solid iron sphere with a diameter of 6cm?
(Note 1: You will need to change both lengths into metres before you do the calculation. Note 2: Your answer will also be the approximate number of iron atoms in a straight line going from one side of the iron sphere to the other. Note 3: This is different to the actual number of atoms in the sphere which is about 8.7 trillion trillion!)
Bonus Feature 1: The Ever-changing Periodic Table
The Periodic Table is of course very familiar to us. However, since 1869, when Dmitri Mendeleev first developed it, the Periodic Table has undergone many, many revisions.
Only 63 elements are shown in this 1871 table, because that’s how many were known at the time, but, by 1902, 80 elements were known. This 1896 version of the Periodic Table shows Helium and Argon here as unclassified elements, because they had only just been discovered, but scientists quickly realized that they were extremely unreactive gases, so a whole new Group, which they first called Group 0, had to be created to accommodate them.
Here’s Group 0 in Mendeleev’s 1905 table. Almost immediately after Helium and Argon were discovered, scientists correctly predicted that more unreactive gases would be discovered which would fill the spaces in the new Group 0, and within a few years, neon, krypton and xenon were also discovered. These gases are now often called the Noble Gases and are in Group 18.
This 1925 Periodic Table was one of the first to split the tall Groups up and to place the transition metals in the middle, so it looks very similar to the form of the Periodic Table that we’re most familiar with today. Soon after it became more common to place the Noble Gases over on the right.
Notice also, that electron configurations for the Noble Gases are also included. These electron configurations had only recently been worked out.
In this table, Argon’s symbol is A. This was commonly used until 1957, when it was officially made Ar.
Up until 1990, the year I graduated from university and started teaching, I don’t know why I grew a beard, the tall Groups of the Periodic Table were numbered from 1 to 8 (often in Roman Numerals but not always) with the Transition metals being in between Groups 2 and 3, and you might come across some Periodic tables that still label the Groups this way. This more closely matches Mendeleev’s original Periodic table from the 1870s. You can see, for example, that Boron and Aluminium were in Group 3 of Mendeleev’s Table, and Carbon and Silicon were in Group 4. (Mendeleev’s Group 8, however, was filled mostly with what we now call Transition Metals.)
This numbering system actually made it fairly easy to remember that the Group 3 elements had three outer-shell electrons and the Group 4 elements had four outer-shell electrons and so on. This was the system I was taught by my high school teachers in the 1980s. However, there was no worldwide agreement about how to number or label the Transition metals and a number of different systems were in use. One system labelled them 3A, 4A and so on, while another system labelled them 3B, 4B and so on. Since there was a little bit of confusion, the so-called International Union of Pure and Applied Chemistry (or IUPAC), an organization made up of scientists from all around the world, in 1990 decided to end the confusion by recommending that the Groups be numbered from 1 to 18.
Now, everyone has to remember that the elements in group 13, or the third tall column, have three electrons in their outer shell, and that the elements in Group 14 (four-teen), or the fourth tall column, have 4 electrons in their outer shell and so on. But that shouldn’t really be a problem!
Many other forms of the Periodic Table have been designed, but the format that has become pretty standard across the world is this one.
So what’s the point of all this? It’s that our knowledge of the natural world is constantly developing, and, as new discoveries are made, we have to be prepared to accept new ways of thinking about things. Never let what you learned yesterday, stop you from learning something new today, because everything’s always changing, and that’ll never change!
(Because change is constant and novelty is nothing new.)
Bonus Feature 2: Flame Tests Practical Activity:
 Rather than using an actual salt in a flame test (here I’m using copper chloride), which can fall off the spatula and make a big mess, it’s much easier to dissolve the salt in water and then let the salt solution soak into an icy-pole stick, well, that’s what I call them. Whenever any salt is dissolved in water it’s called a solution: these are solutions of the chlorides of copper, potassium, barium, lithium, strontium and calcium.
Rather than using an actual salt in a flame test (here I’m using copper chloride), which can fall off the spatula and make a big mess, it’s much easier to dissolve the salt in water and then let the salt solution soak into an icy-pole stick, well, that’s what I call them. Whenever any salt is dissolved in water it’s called a solution: these are solutions of the chlorides of copper, potassium, barium, lithium, strontium and calcium.

 When for example, an icy-pole stick soaked in copper chloride solution is held over the Bunsen burner flame, the copper chloride gets vaporised by the flame and it produces coloured light. No other chemical glows with quite the same colour. The other salts produce different colours. The stick will eventually catch on fire, so it’s best to have a container of water nearby.
When for example, an icy-pole stick soaked in copper chloride solution is held over the Bunsen burner flame, the copper chloride gets vaporised by the flame and it produces coloured light. No other chemical glows with quite the same colour. The other salts produce different colours. The stick will eventually catch on fire, so it’s best to have a container of water nearby.
Now while you can use a spectroscope to examine the emission spectrum from a gas-discharge tube, never use one near an open flame, because you might of course get too close, and you know what might happen.
By the way, the early spectroscopes used a prism to separate the light into its component wavelengths, but modern spectroscopes, even cheaper ones typically found in schools, like this one, use what are called diffraction gratings, which are made of glass or plastic with 1000s of fine lines etched into them.
The way they work is a little complicated, but, like prisms, they too split light into its component colours as you can see here. If we look at a fluorescent light through this spectroscope, we can see that what we think is white light is actually a mixture of different wavelengths. Diffraction gratings had almost completely replaced prisms in spectroscopes by about the late 1800s.
CREDITS
- Thanks to Heinrich Pniok at http://pse-mendelejew.de/en/ for his kind permission in allowing us to use his copyrighted images of the elements. Visit his website to view what is arguably the internet’s best collection of photos of elements. See also http://commons.wikimedia.org/wiki/User:Alchemist-hp?uselang=fr.
- Thanks to the Chemistry Cultural Collection, School of Chemistry, University of Melbourne
- Clip art by Microsoft
- Actinium image used with permission.
- The Ac-225 in the vials was produced and purified by scientific staff at Oak Ridge National Laboratory, and the image was taken by them.
- The funding to produce the Ac-225 was provided by the U.S. Department of Energy (DOE) Isotope Program managed by the Office of Nuclear Physics in the DOE Office of Science.
-
- Cesium.jpg by Dnn87 is licenced under Creative CommonsAttribution-Share Alike 3.0 Unported
- lepidolite-208658.jpg by Rob Lavinsky/ iRocks.com is licensed under Creative Commons Attribution-Share Alike 3.0 Unported.
- CsCrystals1.JPG by Dnn87 is licensed under Creative CommonsAttribution 3.0 Unported .
- RbH.JPG by Dnn87 is licensed under Creative Commons Attribution 3.0 Unported.
- X-ray_spectrometer,_1912 _(9660569929) by Science Museum London is licensed under Creative CommonsAttribution-Share Alike 2.0 Generic
- Atomic Diameter Data in Part E were obtained from the following two sources.
- Clementi, E.; Raimond, D. L.; Reinhardt, W. P. (1967). “Atomic Screening Constants from SCF Functions. II. Atoms with 37 to 86 Electrons”. Journal of Chemical Physics47(4): 1300–1307. Bibcode:.47.1300C. doi:10.1063/1.1712084.
- Slater, J. C. (1964). “Atomic Radii in Crystals”. Journal of Chemical Physics41 (10): 3199-3205. Bibcode:.41.3199S. doi:10.1063/1.1725697.
See http://en.wikipedia.org/wiki/Atomic_radius and http://en.wikipedia.org/wiki/Atomic_radii_of_the_elements_(data_page) for more details.
-
- Steelworks footage © Altos Hornos de Mexico. Used with permission.



